- Worksheets>
- Science>
- Atoms and Molecules
Atoms and Molecules Worksheets
The basic building block of all matter, atoms are at the core of all that we see, touch, smell, feel, and taste. Molecules are formed when two or more atoms link up. A reasonable knowledge of atoms and molecules is indispensable for us to demystify the universe. Both substantially informative and excitingly illustratory, our atoms and molecules worksheets for grade 5 through grade 8 take students through all relevant aspects of the topic with such delicate attention for detail that by the end of the worksheets, learners leave thoroughly educated. Start off with our free worksheets and come back for more!
With concise-yet-telling definitions and easy-to-grasp examples, this printable atoms and molecules chart for grade 5 and grade 6 is a perfect prelude to the topic. The illustrations help ease into the concept.
How are atoms different from molecules? Take the hassle out of reviewing with this pdf to tell the difference between key terms like atoms and molecules, elements and compounds, homogeneous and heterogeneous mixtures, and more.
How good are your students in writing the chemical formulas? Identify each constituent element, write its chemical symbol, and indicate the proportionate number of atoms of each element in this chemical formulas worksheet pdf.
Identify Elements, Compounds & Mixtures
Is water an element, or is it a compound? Don't freak out! Identify substances as elements, compounds, and homogeneous mixtures with this worksheet for 5th grade and 6th grade learners.
Molecules of Elements or Compounds?
Molecules of most elements are made up of two or more atoms of the same element, while compounds are made of atoms of two or more different elements. Cut out the structures, sort, and glue them to distinguish between elements of molecules and elements of compounds.
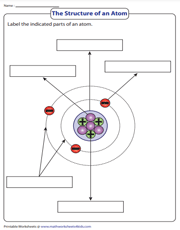
It's high time we dug deep into an atom. In this printable, students broaden their horizons to know the structure of an atom that includes the nucleus, protons, neutrons, electrons, and electron shells.
Continue to excite grade 6 and grade 7 students with the task in this printable worksheet to label various parts of an atom, including the nucleus, a proton, a neutron, an electron.
Matching Structures and Formulas
With two distinct formulas for each compound in action, the interest in this worksheet becomes twofold. 7th grade and 8th grade learners are expected to match the structural formula to the molecular formula.
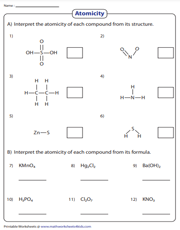
Atomicity is the number of atoms in the molecules of an element. In this 2-part atomicity worksheet, students interpret the atomicity of compounds first from their structures and then from their formulas.
The periodic table is an organized display of all the chemical elements. Go through these printable worksheets and activities that help comprehend the families, and groups of elements in the periodic table.
(5 Worksheets)
Electronic Configuration | Chart
Electron configurations are the summary of where the electrons are around a nucleus. A perfect practice resource, this chart defines electronic configuration and illustrates it using the Aufbau Principle.
Identifying Electronic Configuration
"SPDF" refers to the four different types of orbitals. In this grade 6 and grade 7 worksheet, children prove their mettle by correctly writing the electron configurations of elements using the SPDF notation.
Identifying Elements Based on Electronic Configuration
Watch the SPDF notation at play yet again! In this pdf worksheet, though, the task for students is a little different. Let the young scientists identify the elements denoted by the given electronic configurations.
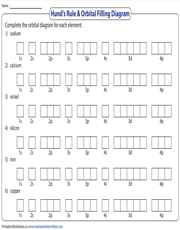
Hund's Rule | Orbital Filling Diagram
Hund's Rule clearly defines the behavior of unpaired valence shell electrons. In this printable exercise with six elements, high school students are tasked with drawing the Hund's Rule orbital diagram for each element.
Both transferable and shareable, valence electrons are the electrons in the outermost shell of an atom. In this pdf, middle school students write the electron configuration and write the valence electrons of each element.
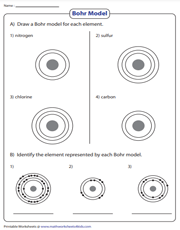
Proposed by the Danish physicist Niels Bohr, the Bohr model is a household name in Chemistry. Children in grade 6, grade 7, and grade 8 draw a Bohr model for each element and identify the element represented by each Bohr Model.
A Lewis structure is one where element symbols represent atoms, and dots represent their electrons. In this worksheet, students draw the Lewis dot structure for each element, molecule, and compound.
The questions in this printable exercise include defining the laws of conversation of mass and constant proportions, explaining the two types of ions, and telling between isotopes and isobars.